Biocatalysis
Mechanism + Description
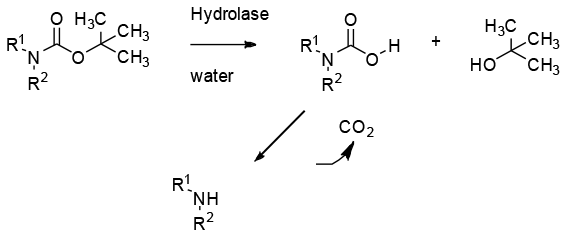
In general, via the normal mechanism of ester/amide hydrolysis used by hydrolytic enzymes.
General comments
Potentially a very green route to deprotect BOC protected amines and also generating t-BuOH as a product avoiding any issues with the generation of t-Bu cations. However, BOC is very hindered and the carbamate group can act as an inhibitor towards commonly used lipases and esterases used in organic synthesis. Other carbamates like CBZ are more likely to undergo enzyme –catalzsed hydrolysis. Whilst currently a rare and under utilized transformation, some research groups are looking at developing BOC hydrolase enzymes.
Key references
Relevant scale up example
None located.