Heterogeneous Catalysts
Mechanism + Description
A number of heterogeneous solid catalysts have been reported to cleave BOC groups. These are mostly sulfonated resins in their acid forms, although other solids have been reported to cleave BOC groups.The reaction is probably catalyzed by acidic sites on the solid surface, so all heterogeneous catalysts most likely follow the acid cleavage mechanism.
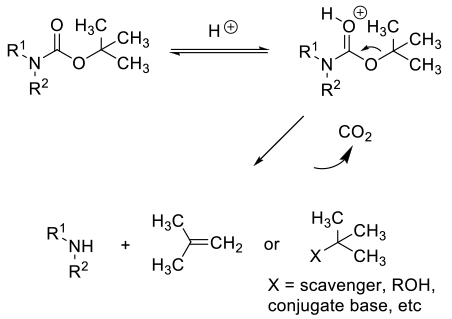
General comments
BOC removal with heterogeneous catalysts can be beneficial if an aqueous work-up to remove soluble acids needs to be avoided, since after deprotection the amine product will form a salt with acid functions on the solid, at least a molar
equivalent of acid catalyst needs to be used. This can lead to quite high weight equivalents of solid catalyst that need to be charged. After reaction, the amine product will be bound to the resin and as an ion exchange separation, will need to be eluted. Some heterogeneous catalysts show good selectivity for removing BOC groups on aromatic amines in the presence of BOC protected alkyl amines. If synthetic polymers are used in c-GMP synthesis, appropriate attention needs to be made to leachables.
Key references
Adv. Synth. Catal. 2003, 345, 1207-1208 Selective Removal of N-Boc Protecting Group from Aromatic Amines Using Silica Gel-Supported Sodium Hydrogen Sulfate and HY-Zeolite as Heterogeneous Catalysts
J. Org. Chem, 2005, 9 (70) 3495 – 3499 BOC removal with SiO2
Tet. letts. 2000, 41, 385-387 A mild procedure for the clay (K10) catalyzed selective removal of the tert-butoxycarbonyl protecting group from aromatic amines
Relevant scale up example
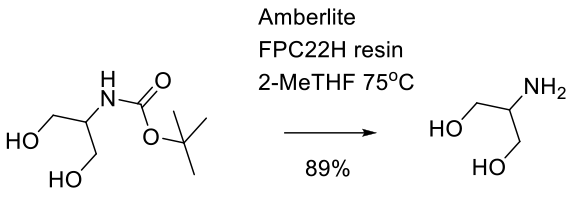
Experimental
400 g scale
Org. Process Res. Dev. 2009, 13, 478–482