Tin Reagents
Mechanism + Description
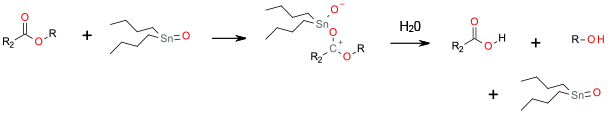
Coordination of tin to the ester activates towards nucleophilic attack by water.
General comments
Bis(tributyltin)oxide, (BBTO) acts as a mild, neutral, and chemoselective ester cleavage agent for a variety of carboxylic esters and thioesters with good to excellent yields. BBTO may be useful when it is necessary or desirable to avoid exposing a particular substrate to acidic, basic or hydrolytic conditions. This has particular applicability for natural products where chemo- and regioselective deprotection of the carboxyl group under exceptionally mild conditions is required, e.g. b-lactam, carbohydrate and peptide chemistry. Organotin compounds are persistent and toxic with high impact on human health and the environment .Most organotin compounds are endocrine disruptors and on the REACH SVHC list, to be phased out in the EU by 2015. Contamination of equipment and cleaning maybe also an issue.
Key references
J. Org. Chem., 1994, 59, 7259 Scope / Mechanism of Deprotection of Esters by Bis(tributyltin) Oxide
J. Chem. Soc. Perkin Trans. 1, 1996, 99 Selective deprotection of phenacyl, benzyl and methyl esters of N-protected amino acids with bis(tributyltin) oxide
Synlett, 2000, 140 [tBu2SnOH(Cl)]2 as a Highly Efficient Catalyst for Deacetylation
Relevant scale up example
None found