TMS Iodide
Mechanism + Description
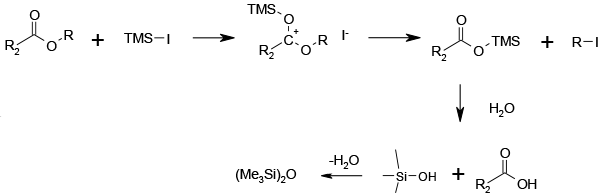
TMS acts as a Lewis acid activating the ester alkyl group to SN2 attack by iodide. This generates an Alkyl iodide and the corresponding TMS ester which is hydrolysed by water to the acid and (Me3Si)2O.
General comments
Trimethylsilyl iodide can be used directly or prepared in situ from trimethylsilyl chloride and sodium iodide. The reaction is frequently carried out in acetonitrile at, or close to, ambient temperature although other solvents (notably chlorinated solvents, though their use is discouraged) have been used at higher temperatures. Upon aqueous drown out the free acid is obtained in good to excellent yields. This reagent combination can also be used to cleave ethers, convert alcohols to iodides, convert acetals to the corresponding aldehyde or ketone, and reduce sulphoxides to sulphides. The reagent has also been used to remove N-benzoyl and N-Boc in peptides. TMSI itself is prone to hydrolysis and it is unstable to light. Therefore it is recommended that it be prepared in-situ, either using the method above or through the treatment of Hexamethyldisilane with iodine. Upon aqueous work up, (Me3Si)2O is formed as a by-product.
Key references
J. Org. Chem., 1983, 48, 3667 Synthetic transformations with trichloromethylsilane/sodium iodide reagent
J. Am. Chem. Soc., 1977, 99, 968 Quantitative dealkylation of alkyl esters via treatment with trimethylsilyl iodide
Chem. Commun., 1978, 874 dealkylation of esters, ethers, and acetals by chlorotrimethylsilane–sodium iodide
Synthesis, 1977, 417 Mild and effective cleavage of esters and ethers with phenyltrimethylsilane/iodine
Angew. Chem. Int. Ed., 1979, 18, 612 Hexamethyldisilane/iodine: convenient in situ generation of iodotrimethylsilane
Relevant scale up example
None found
Green Review
-
Atom efficiency (by-products Mwt)
Poor atom economy – dealkylation generates TMS2O ( 162) after quench and the corresponding alkyl iodide. - Safety Concerns
None apparent. TMSI is corrosive and reacts with water. TMSI may be incompatible with certain solvents e.g. ethers. - Toxicity and environmental/aquatic impact
Organoiodine compounds, especially hydrophobic materials, present a moderate hazard to the aquatic environment and may bioaccumulate. TMS2O will not biodegrade. Alkyl iodide by-products may present PGI issues, and volatile organoiodides like MeI may need off-gas control. Possible issues with any incineration of waste solvents. - Cost, availability & sustainable feedstocks
Generally a high cost reagent for organic synthesis on large scale. If prepared from TMSCl and NaI the cost is greatly reduced as are stability issues upon storage. - Sustainable implications
Incineration of waste streams could be problematic (iodine content). Limited utility for waste by-products. Iodine is an element at medium to high risk of depletion. High LCA reagent, although it is possible to recover iodide from waste materials.