TEMPO-Hypervalent Iodine Oxidation
Mechanism + Description
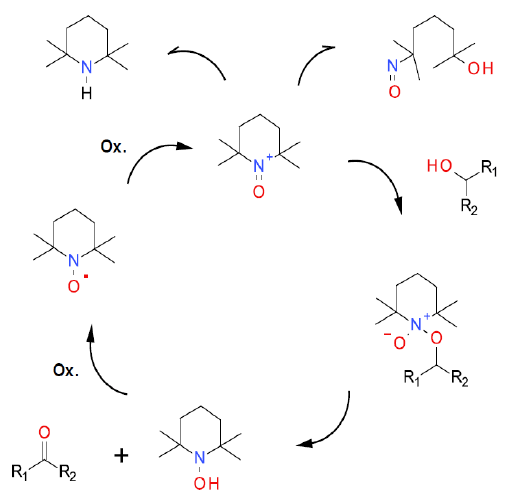 The hypervalent iodine reagent is present to act solely as the reoxidant for the N-hydroxy intermediate, so the mechanism is as the same as any TEMPO oxidation. Hypervalent iodine reagents can be used for the direct oxidation of alcohols, but these are IV and not IIII.
The hypervalent iodine reagent is present to act solely as the reoxidant for the N-hydroxy intermediate, so the mechanism is as the same as any TEMPO oxidation. Hypervalent iodine reagents can be used for the direct oxidation of alcohols, but these are IV and not IIII.
General comments
(Diacetoxyiodo)benzene is routinely used as the hypervalent iodine-oxidizing partner to TEMPO. As with other TEMPO/oxidizing mixtures, rate increase can be facilitated by phase transfer catalysis. One benefit compared to other re-oxidants is that reactions can be run under totally anhydrous conditions.
Key references
Ambreen, N.; Kumar, R.; Wirth, T. Hypervalent iodine/TEMPO-mediated oxidation in flow systems: a fast and efficient protocol for alcohol oxidation. Beilstein J. Org. Chem. 2013, 9, 1437-1442.
Li, X.; Zhang, C. An Environmentally Benign TEMPO-Catalyzed Efficient Alcohol Oxidation System with a Recyclable Hypervalent Iodine(III) Reagent and Its Facile Preparation. Synthesis. 2009, 7, 1163-1169.
Zhdankin, V. V. Hypervalent iodine(III) reagents in organic synthesis. ARKIVOC. 2009, i, 1-62.
Relevant scale-up example
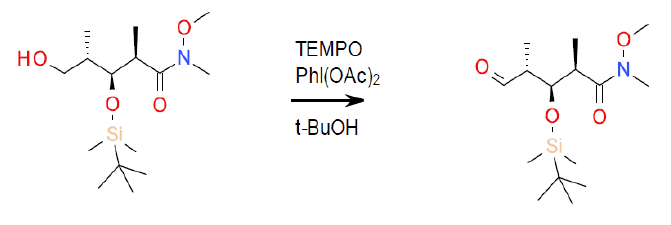
Org. Process Res. Dev. 2004, 8 (1), 101–106. – 50-L scale oxidation.

Org. Process Res. Dev. 2006, 10, 814–821. – 100-L prep in DCM
Green Review
- Atom efficiency (by-products)
Poor atom economy – removal of H2 generates 324 of waste. Some hypervalent iodine oxidants can be recovered and reused, which would improve poor atom efficiency. - Safety concerns
Low concerns for reagents; all TEMPO oxidations are exothermic and may present delayed exotherms. - Toxicity and environmental/aquatic impact
Organoiodine compounds, especially hydrophobic materials, present a moderate hazard to the aquatic environment and may bio accumulate. Iodobenzene is generated as a by-product. - Cost, availability & sustainable feedstocks
Generally a high cost reagent for organic synthesis on large scale; prepared from petroleum based feedstocks. - Sustainable implications
Incineration of waste streams could be problematic (iodine content). Limited utility for waste byproducts. Iodine is an element at medium to high risk of depletion, although it is possible to recover iodide from organoiodine waste materials.