Biocatalysis
Mechanism + Description
Oxidation of primary alcohols to carboxylic acids occurs via the aldehyde, which is produced via an alcohol dehydrogenase or alcohol oxidase, followed by an oxidation by aldehyde oxidase. Rarely, the aldehyde hydrate can be oxidized with a dehydrogenase. All the enzymes need a terminal electron acceptor – either NAD(P)+ for dehydrogenases or O2 for oxidases.
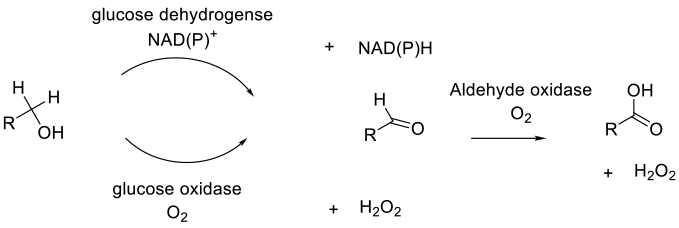
General comments
There are few enzymes that can directly catalyze the oxidation of a primary alcohol to a carboxylic acid. In most cases, two enzymes are required: one to oxidize to the aldehyde and a second to oxidize the aldehyde to the required acid product. Biocatalytic oxidation is generally very safe, occurring in water or water-solvent mixtures at near ambient temperatures. Reagents are formed from natural sources and are readily degradeable. Issues with exotherms and delayed exotherms often encountered in chemical oxidations are absent, though if air needs to be supplied as a terminal oxidant, care needs to be taken in generating flammable mixtures if organic solvents are present. Oxidases generate peroxide as a by-product, and this often needs to be removed by the addition of a catalase since most enzymes are deactivated by high concentrations of peroxide.
Key references
A review of biocatalytic oxidation of primary and secondary alcohols: Romano, D.; Villa, R.; Molinari, F. Preparative Biotransformations: Oxidation of Alcohols. ChemCatChem. 2012, 4 (6), 739-749.
Relevant Scale-Up Examples with Scheme
None found
Green Review
-
Atom efficiency (by-products Mwt)
Atom efficiency depends on the terminal oxidant and any co-factor recycling required. Oxidases using O2 give H20 as the by-product. Dehydrogenases need co-factor recycle. If using whole cells, this may be possible through cell metabolism. If not, other enzyme and/or chemical oxidants can be employed; ideally, the choice should be made so as to minimize any mass burden on the process. - Safety Concerns
Generally, biocatalytic methods are free of thermal events and can be managed in standard equipment. - Toxicity and environmental/aquatic impact
The biocatalysts are commonly biodegradable and pose minimal environmental hazards. Some of the proteins, especially dusty solids, can be sensitizing (R42), so appropriate handling should be employed. For use in the c-GMP manufacture of an API, enzymes from mammalian sources or those fermented using mammalian products should be avoided. Older work using whole cells and biphasic systems often use dialkyl phthalates as the organic phase. These materials are persistent and endocrine disruptors, hence they should be avoided. - Cost, availability & sustainable feedstocks
Many enzymes are now becoming commercially available in bulk, and many CROs can offer biocatalysis and enzyme development services. In most cases, at the pilot stage, a biocatalyzed reaction will cost more than a chemical alternative (fermentation/enzyme supply is very sensitive to economies of scale), but at full scale, most biocatalyzed processes are greener and cheaper than their chemical alternatives. - Sustainable implications
Enzymes are produced from renewable materials and are fully biodegradable back to innocuous natural products (amino acids). The use of modern molecular biology and fermentation technology has greatly reduced the LCI of enzyme manufacture. Maximum sustainability benefits are usually obtained with mutant recombinant enzymes rather than natural enzymes.